Primary Immunodeficiencies
Immune Monitoring
Last update: October 1st, 2019
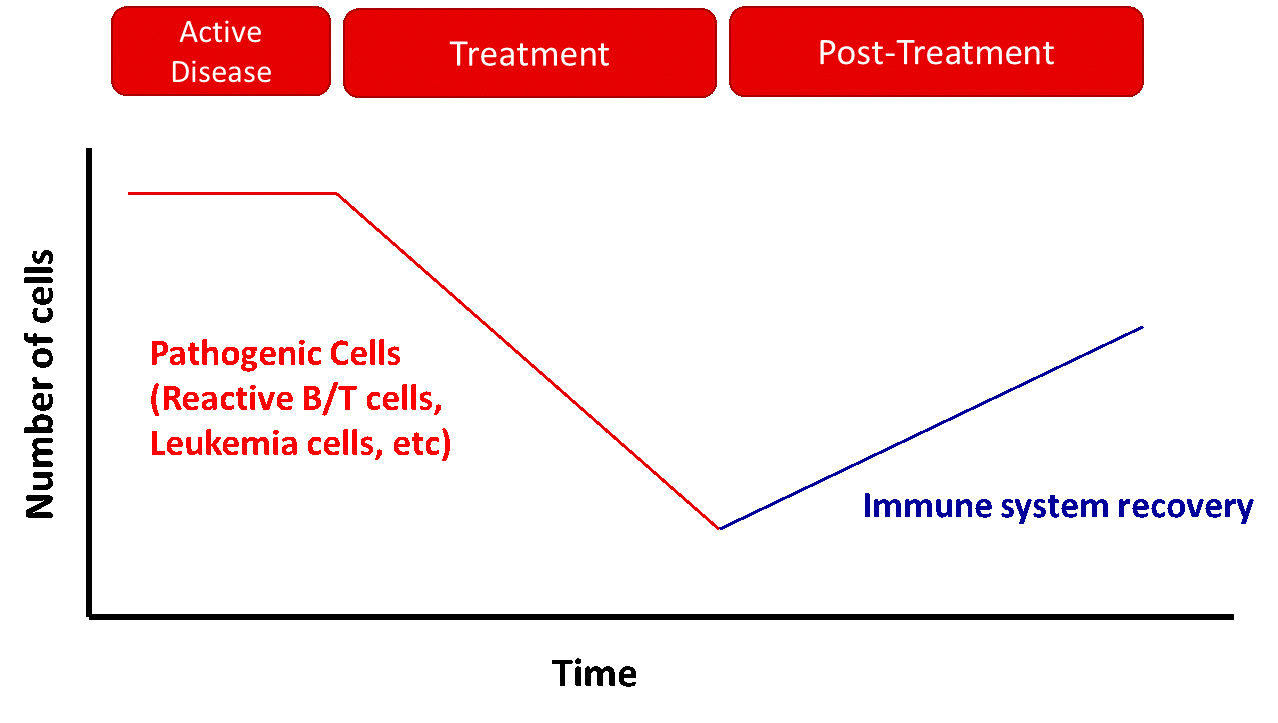
Immune monitoring is the study of one or several populations of the immune system over time. It is useful to evaluate immune populations in response to the treatment of a certain disease or to follow up the immune reconstitution after the immune system depletion. Recent studies highlight the importance of the assessing not only the pathological population, but also the immune context as it may give very relevant information about the development of the disease such as the level of the pathological population, unveil prognostic markers which in the future will allow for better clinical decisions.
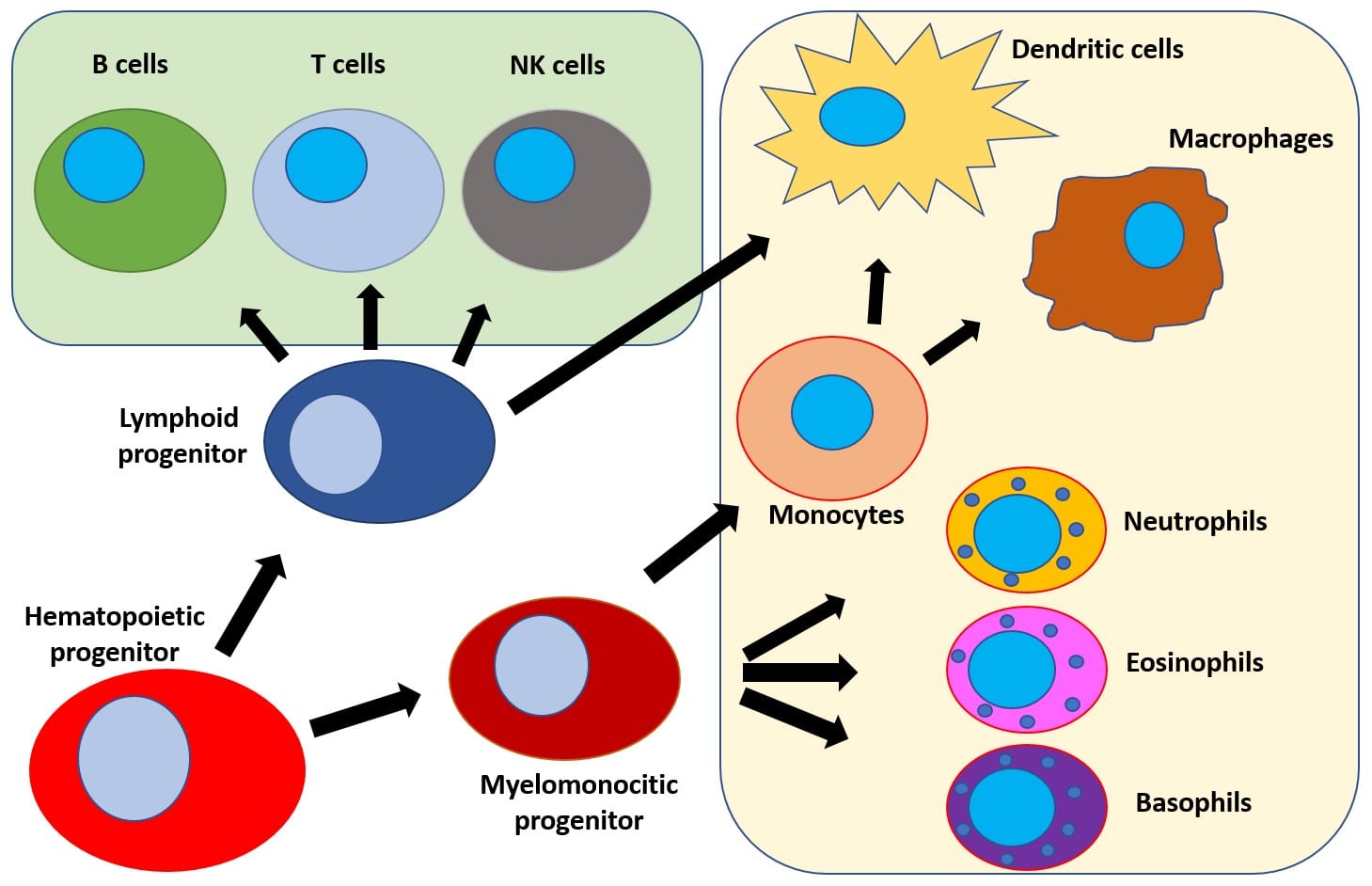
Assessing the different stages of B-cell maturation allows to follow up diseases with a demonstrated B-cell compartment implication, such as most auto-immune diseases and B-cell lymphoproliferative disorders. The T-cell subpopulation give indication on the behavior of memory and effector populations over time. The overall recovery of immune system homeostasis can be studied which may be especially relevant in fields such as oncology, auto-immune diseases or in the response to vaccination and to advance treatment and management of many of these diseases.
Resources
Publications:
- Maecker HT. (2006) The Role of Immune Monitoring in Evaluating Cancer Immunotherapy. In: Disis M.L. (eds) Immunotherapy of Cancer. Cancer Drug Discovery and Development. Humana Press. Go to publication
- Hegde PS et al. The Where, the When, and the How of Immune Monitoring for Cancer Immunotherapies in the Era of Checkpoint Inhibition. Clin Cancer Res. 2016;22(8):1865-74. Go to publication
- Justyna O, et al. Immune Reconstitution after Allogeneic Hematopoietic Stem Cell Transplantation. Frontiers in Immunology. 2016;7:507. Go to publication
- Scutti JA, Travassos LR and Vence LM. (2018). Immune Checkpoint Blockade and Immune Monitoring. Immunoregulatory Aspects of Immunotherapy. Seyyed Shamsadin Athari. IntechOpen. Go to publication