The flow cytometer
Acoustic focusing
Last update: October 1st, 2019
Flow cytometry data quality depends on collecting information from thousands of individual cells as they pass through the laser interrogation point. In conventional flow cytometry, sheath fluid hydrodynamically focuses cell or particle suspensions into a narrow sample stream. In combination with a tightly focused laser, this narrow sample stream creates a small interrogation volume that is analyzed via high numerical aperture optics. The tight positioning ensures highly precise analysis and the small interrogation volume enables largely homogeneous assays (wash-free). However, the analysis speed of conventional cytometers is not enough to support rare event detection applications that have clinical relevance (e.g. detection of circulating tumor cells present in blood at levels as low as a 100 cells per milliliter).
Higher analysis rates, limited in conventional flow cytometry, depend on several parameters that include the maximum analysis rate that is determined by the stochastic nature of cellular arrival at the interrogation volume, and limiting the concentrations of cells that can be used without causing an intolerable number of coincidences following the Poisson distribution of particle arrival times. Acoustic positioning of the cells has been developed and have the advantage of concentrating particles to precise positions without the concurrent acceleration imparted by hydrodynamic focusing, which allows for high particle analysis rates at reduced linear velocities. Furthermore, these techniques do not require a sheath flow, thus fluid consumption and hazardous waste output is minimized.
The acoustic technology generates ultrasonic waves that act to transport particles to the center of the sample stream (acoustic focusing). The pre-focused stream is injected into the sheath stream, which applies hydrodynamic pressure to the sample (traditional hydrodynamic focusing). The combination of these two forces results in a narrow core stream and an increased probability that cells are passing single file through optical system.
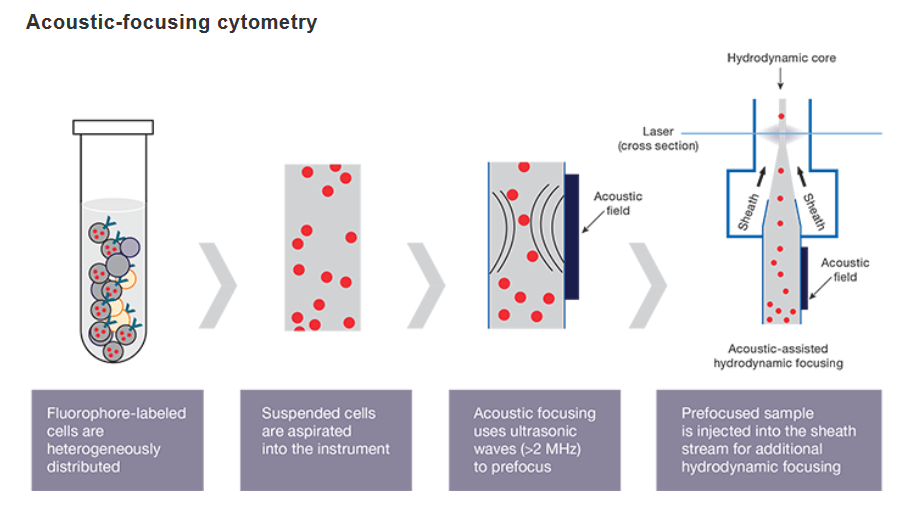
Acoustic focusing positions biological cells and other particles over a wide range of sizes, can utilize wide channels, and supports high volumetric flow rates for flow cytometry.
Flow cytometers equipped with acoustic-assisted hydrodynamic focusing, are able to process samples faster than traditional cytometers. This breakthrough technology uses sound waves for more precise cell alignment and higher data quality. The pre-alignment of cells allows to process samples quickly and acquires more statistically significant events even from very dilute samples.
Resources
Publications:
- Adan A, et al. Flow cytometry: basic principles and applications. Crit Rev Biotechnol. 2017 Mar;37(2):163-176. Go to publication
- Cossarizza A, et al. Guidelines for the use of flow cytometry and cell sorting in immunological studies. Eur J Immunol. 2017 Oct;47(10):1584-1797. Go to publication
- Piyasena ME, et al. Multinode acoustic focusing for parallel flow cytometry. Anal Chem. 2012 Feb; 84(4): 1831–1839. Go to publication
- Ward M, et al. Fundamentals of Acoustic Cytometry. Curr. Protoc. Cytom. 49:1.22.1-1.22.12. Go to publication
- Brown M and Wittwer C. Flow Cytometry: Principles and Clinical Applications in Hematology. Clinical Chemistry. 2000 April;46:8(B)1221–1229. Go to publication
- Howard M. Shapiro Practical flow cytometry, 4th ed. John Wiley hi Sons, Inc. 2003 August; 736 Pages. Go to publication

