Multiple Myeloma
The number of immunophenotypic markers that can be evaluated in 8-color assays and the high number of cells interrogated using the MM MRD methodology increase the complexity of data analysis. EuroFlow™ developed and validated a reference database for MRD assessment in bone marrow samples after treatment. The database should only be used with files which follow the EuroFlow™ standardized operating procedures.2
How was the MM MRD database built?
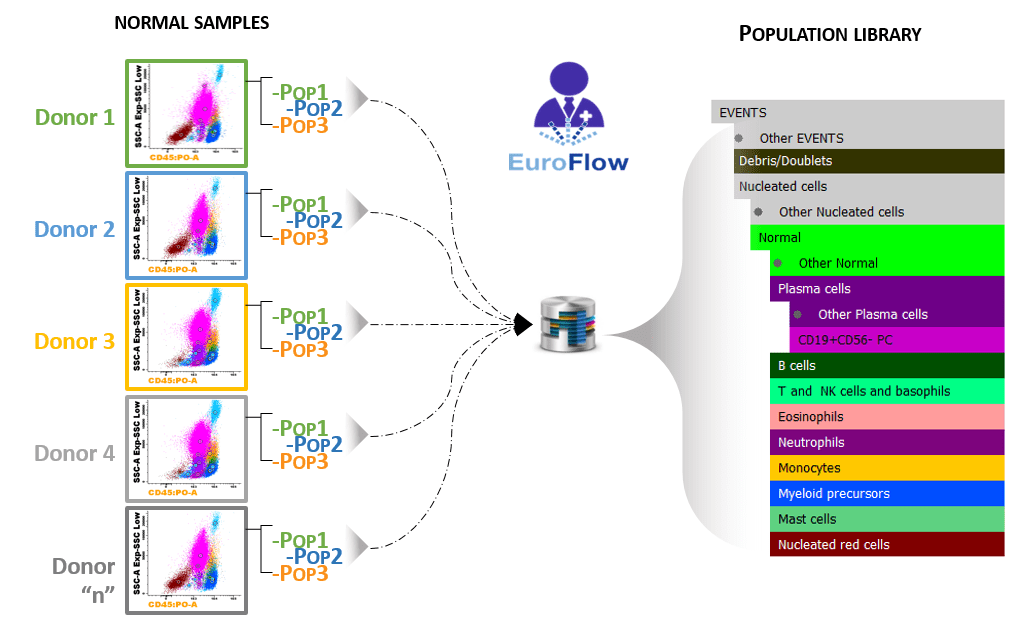
EuroFlow™ developed and validated a database containing representative bone marrow samples from normal/healthy donors stained with the MM MRD panel following EuroFlow™ SOPs.
In MM MRD Database, the samples were collected in different EuroFlow™ centers to reflect biological and technical variability regarding different laboratories, instruments and operators. EuroFlow™ experts analyzed those samples and created a library of reference populations. When comparing a new sample with this database, all populations of the sample will be automatically identified.2
How to compare a new case with EuroFlow™ MM MRD Database using Infinicyt™ Automated Gating & Identification tool?
- Compatible FCS file
Acquired using the EuroFlow™ SOPs for instrument set-up, sample preparation and antibody panel. - Clustering
All events are automatically identified by clustering algorithms. - Identification with EuroFlow Databases
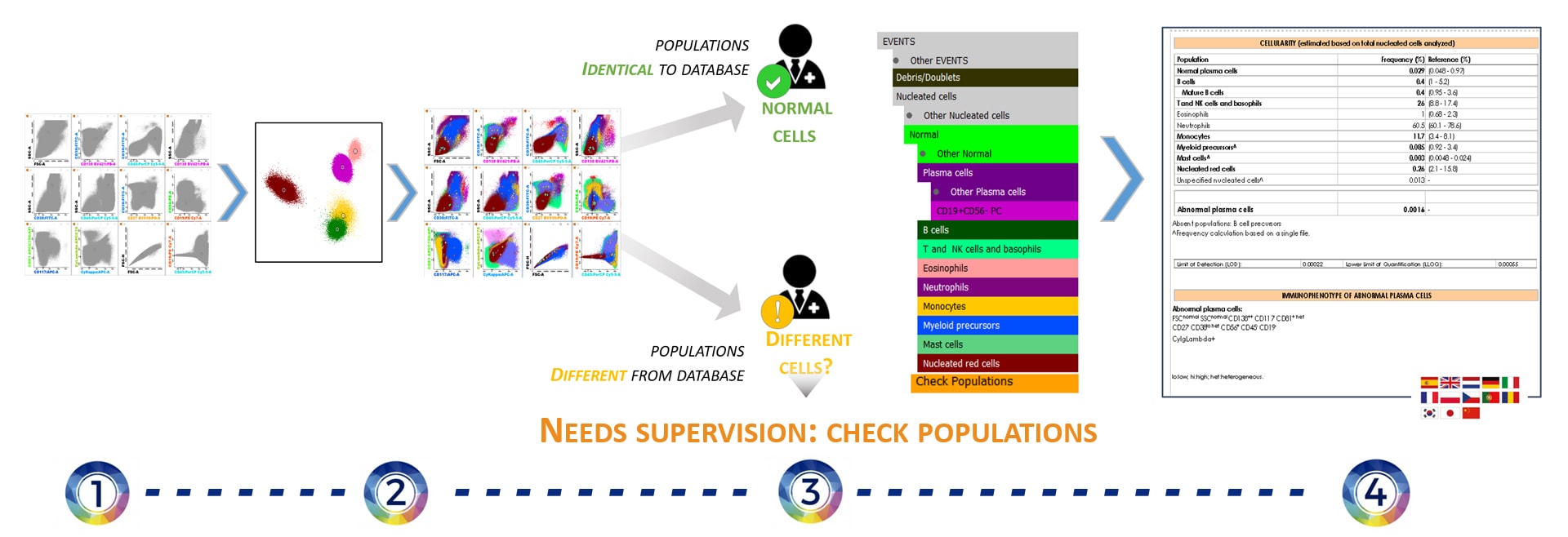
Infinicyt™ automatically compares the clusters with EuroFlow™ Database:
– Clusters identical to cell populations included in the database are directly renamed with the name of those populations.
– Clusters that are not identical to the ones included in the database are assigned to Check Populations. The user has to review them and decide whether they are normal or abnormal. - Automatic Report includes:
– Frequency of each population with reference ranges.
– Relevant information for clinical routine automatically updated for each sample.
– Available in 13 different languages.
Click to see and example of MM MRD Report.

Resources
Publications:
- Flores-Montero, et al. Immunophenotype of normal vs. myeloma plasma cells: Toward antibody panel specifications for MRD detection in multiple myeloma. Cytometry B Clin Cytom. 2016 Jan;90(1):61-72. Go to publication.
- Flores-Montero J, et al. Next Generation Flow for highly sensitive and standardized detection of minimal residual disease in multiple myeloma. Leukemia. 2017 Oct;31(10):2094-103. Go to publication.