Clinical Immunology
Due to the success of the standardization in hemato-oncology field, the EuroFlow™ consortium has now expanded their activities to the immunology field. Although quite recent, the NGF methodology for primary immunodeficiencies (PID) has already made available standardized protocols, panels and reference databases (included in the Infinicyt™ Software).
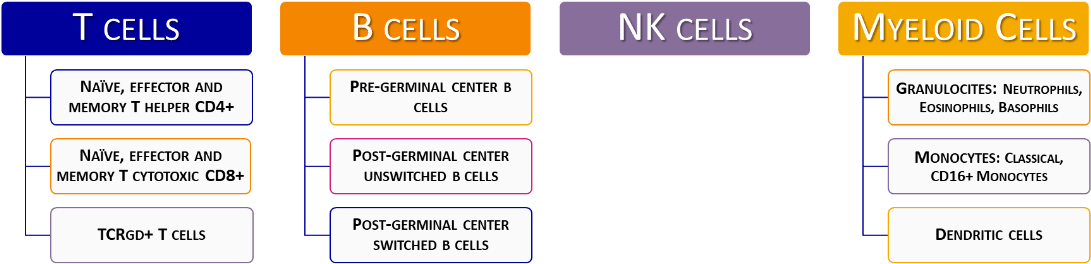
As defined by the ESID registry, the complete diagnosis and classification of PIDs is based on a stepwise screening step and subsequent characterization for numerical alterations in lymphocyte T, B and NK subpopulations, absence of specific proteins and functional assays (www.esid.org).
EuroFlow™ objective was to develop a universal and highly sensitive method to identify all lymphoid PIDs (around 70% of all diagnosed PIDs) which could guide and help prioritize further diagnosis using other techniques (e.g. genetic analysis) (13).
This method consists on the application of the EuroFlow™ methodology (standardized panel, SOPs, etc.) to create a database in the Infinicyt™ Software that describes the frequency and absolute counts of all cell populations that can be identified with the PIDOT tube.
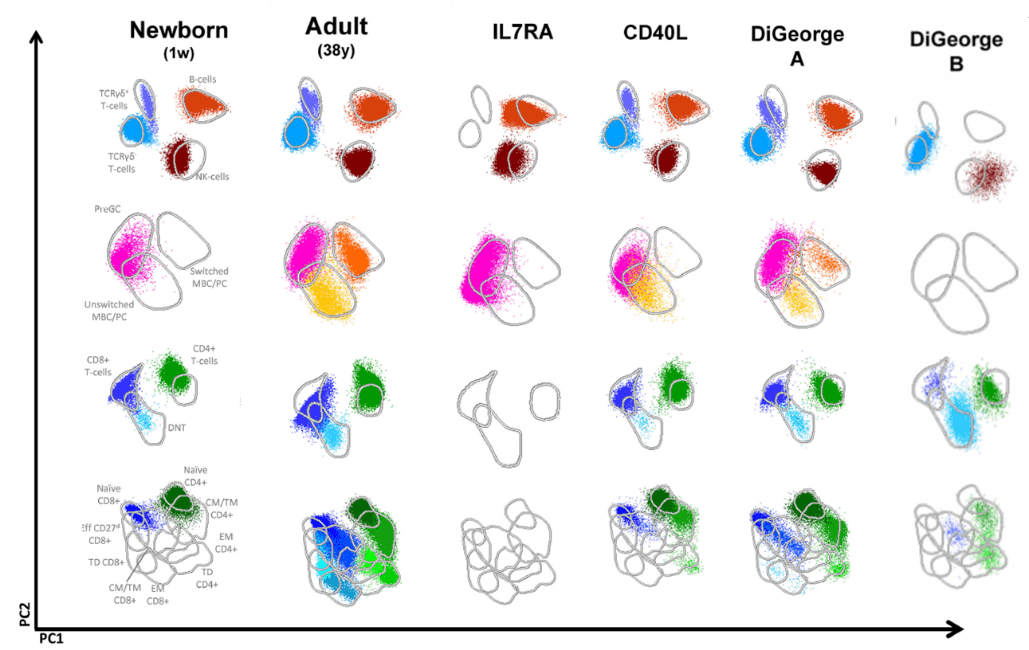
This standardized NGF PID method has been validated on hundreds of healthy controls and PID patients with defined genetic defects and 14 different age-ranges. The frequency and absolute counts were obtained for all normal samples and age groups and included in a robust database (13).
The EuroFlow™ PID database was included in the Infinicyt™ Software. Furthermore, Cytognos and EuroFlow™ developed tools for multidimensional pattern recognition of the maturation pathway of all lymphoid populations to better detect possible alterations. These Infinicyt™ tools intend to overcome the difficulties that so far have complicated PID diagnosis, such as low number of cases per center and lack of standardization between different laboratories.
The major advantage of the standardized NGF PID approach is that results can be fully exchanged between different laboratories in any country of the world and serve as templates to increase global clinical knowledge and also improve current disease registries.